Batteries can be compared to fuel tanks that store energy, where their capacity to hold energy decreases over time. A fuel tank or battery that could hold 100 liters in year 1 may only be able to hold 75 litres in year 10. The rate capacity loss may be slow at first, but it will increase as the tank or battery deteriorates. As the battery loses capacity, it will become less effective at holding or storing the desired amount of energy. Eventually, the battery will become unusable and will need to be replaced.
Battery degradation refers to this gradual loss of capacity and performance of a battery over time. It can occur due to a variety of factors, including chemical and electrochemical reactions within the battery. Whether it be a Loss of Lithium Inventory (LLI) – namely a loss of usable lithium – or a Loss of Active Material (LAM), these ageing mechanisms eventually lead to:
- Decrease in capacity and energy ;
- Increase in internal resistance associated with a decrease in energy and power output.
Additionally, usage pattern and environmental factors can contribute to accelerating these phenomena. In calendar usage profiles, uncontrolled State-of-Charge (SoC) levels and temperature variations significantly contribute to accelerated battery degradation. Similarly, in cycling modes, charging and discharging rates, as well depth of discharge (DoD) and temperature, can have a detrimental effect on battery ageing.
Regarding environmental conditions, high temperatures can, on the one hand, speed up the degradation of the electrodes and electrolyte. On the other hand, cold temperatures can result in temporarily lowering the battery’s performance and participate to irreversible lithium metallic deposition.
Finally, it is important to note that battery chemistry and battery design, at the cell, module, or pack level, can also impact ageing. This is illustrated by the abnormal cell thermal heating observed in some battery design or mechanical pressures experienced by pouch cells.
The monitoring of battery degradation hence requires comprehensive analysis of various factors, making it a challenging task.
Battery irreversible degradation
Ageing mechanisms can be categorized in two main phenomena:
LAM (Loss of Active Material)
LAM refers to the reduction in the amount of active material present in the electrodes of the battery. As described above, this can occur due to a variety of reasons, including over-charging, over-discharging, high temperatures, and long-term use.
When active material is lost, the battery’s capacity and performance will decrease, leading to shorter runtimes and less efficient charging. It is important to note that the active material can be independently lost from the positive or negative electrode.
LLI (Loss of Lithium Inventory)
LLI refers to the reduction in the amount of usable lithium present in the electrodes of a lithium-ion battery. It is mainly occurring in the interface of the negative electrode and electrolyte due either to Solid Electrolyte Interface (SEI) growth or Lithium metallic deposition.
By allocating the total capacity loss of a battery between LAM & LLI – see figure 1 –, Battery Insight® indicators are not only able to accurately monitor its degradation evolution but also detect Lithium metallic deposition before it gets hazardous.
Learn more on battery safety issues: Predictive safety management
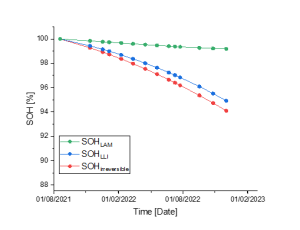
Figure 1. Total irreversible capacity losses and distribution of these losses between LLI and LAM
But then what is SEI?
The growth of the SEI layer can be considered as a parasitic reaction that mainly occurs at the interface between the graphite negative electrode and the electrolyte. This SEI formation leads to a depletion of Lithium ions at the interface, causing a difference in the lithiation level between the negative electrode and the positive electrode, resulting in a loss of cycling Lithium and a decrease in the State-of-Health (SoH) of the battery.
SEI growth can be thus perceived as an “electrode imbalance**” issue!
Battery reversible degradation
In addition to the aging mechanisms and regardless of the SoH, cell imbalances at the module level can also affect the available capacity, i.e. battery State-of-Health (SoH), depending on the usage profile.
Indeed, significant cell imbalance can result in a loss of the available capacity due to one string of cells halting the charge before the rest of the pack is fully charged or stopping the discharge before the rest of the pack is fully discharged. Yet, a rebalancing procedure can fully restore the observed capacity/energy losses.
However, the BMS must be able to detect the imbalance problem, which is problematic for instance in the case of LFP. In fact, BMS relies on voltage delta to detect imbalances, which is not applicable to the voltage plateau based on the charge state of LFP.
Learn more on balancing issues: Zoom on cell-imbalance
It is hence crucial to be able to detect cell-imbalance and distinguish this additional but reversible capacity fade due to BMS cell balancing failure from irreversible battery degradation due to usage conditions.
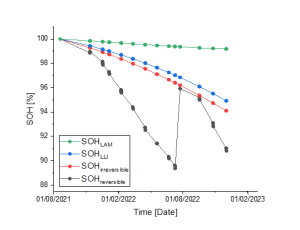
Figure 2. Reversible capacity evolution before and after a cell-balancing procedure
Figure 2. above distinguishes battery degradation due to irreversible aging mechanism – LLI & LAM – from reversible battery degradation due to cell-imbalance in an eBus use-case in operation. We can clearly see a sudden drop in reversible SOH before a cell-balancing procedure around July 2022 that allows to get back 4% SOH. The curve collapses again after a few months due to further cell-imbalance issues. In that context, cell-balancing procedures must take place more often to make the most of battery capacity and extend in some cases its life span.
All in all,
- Battery degradation is either due to irreversible ageing mechanisms – Loss of Active Material (LAM) & Loss of Lithium Inventory (LLI) – caused or accelerated by usage patterns and environmental conditions; or due to reversible cell-imbalance.
- Allocating the total capacity loss of a battery between LAM & LLI is crucial. Not only to accurately monitor battery degradation evolution but also to detect Lithium metallic deposition before it gets hazardous and leads to battery fire.
- Distinguish reversible battery degradation allows to establish cell-balancing procedures make the most of battery capacity and extend in some cases its life span.
- Monitoring LLI is key to evaluate a battery second life: how available is the lithium inventory? What is the level of Lithium metallic deposition and how safe it is to keep using the battery?
**By utilizing a third electrode, it is possible to counterbalance the “electrode imbalance” by charging only the negative electrode without altering the lithiation level of the positive electrode. This method has been proven successful by recovering 100% of an LLI of 20% or more, but it must be noted that this approach is not currently feasible for commercial cells as they do not have a third electrode or a BMS capable of performing the procedure. Thus, the growth of SEI can be considered a permanent occurrence in practical application






